 
Trial RegistrationĬ identifier, NCT02946931.ĭabigatran etexilate (dabigatran) is an oral thrombin inhibitor approved for the prevention of stroke in patients with nonvalvular atrial fibrillation. The results of this interim analysis suggest that idarucizumab is safe and effective for the reversal of dabigatran in Japanese patients in a real-world setting, and support the continued use of idarucizumab. In Group B, normal intraoperative hemostasis was reported in 63 patients (72.4%). In Group A, the median time to bleeding cessation in patients without intracranial bleeding was 3.3 h. The reversal effect of idarucizumab based on aPTT within 4 h after idarucizumab administration was assessed in 30 patients and the median maximum percentage reversal was 100%. 
Eighteen patients (6.9%) experienced ADRs within 4 weeks. This interim analysis included 262 patients who received idarucizumab. The secondary endpoint was the maximum extent of reversal of the anticoagulant effect of dabigatran based on activated partial thromboplastin time (aPTT) within 4 h after idarucizumab administration. The primary endpoint was the incidence of adverse drug reactions (ADRs). 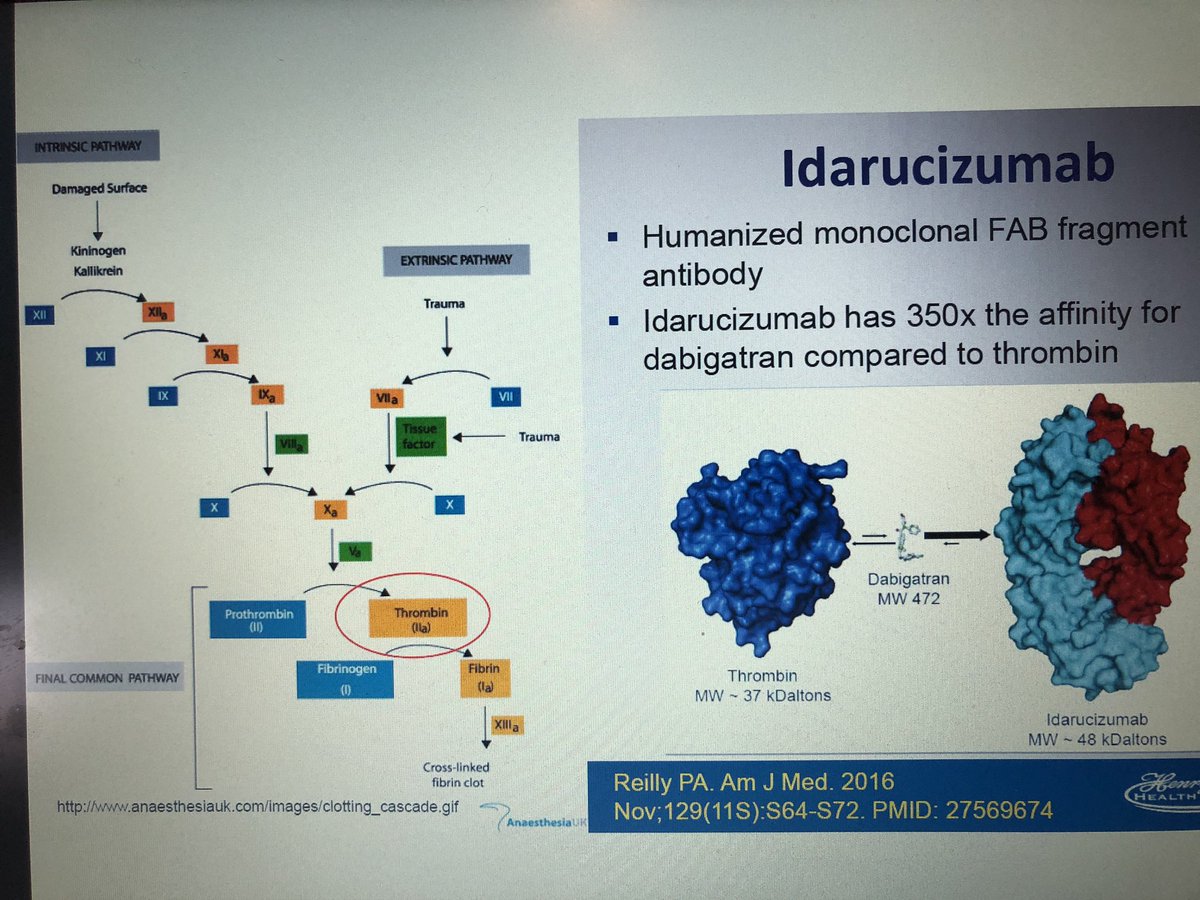
An all-case post-marketing surveillance is ongoing to collect data in Japanese patients treated with idarucizumab who had serious bleeding (Group A) or required an urgent procedure (Group B). Idarucizumab, a monoclonal antibody fragment, was developed to reverse the anticoagulant effect of dabigatran, and it was approved in Japan in September 2016. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
